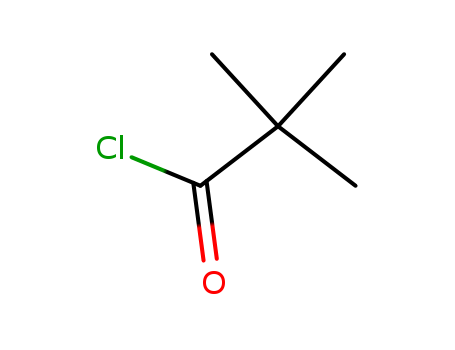
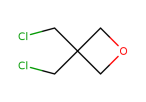
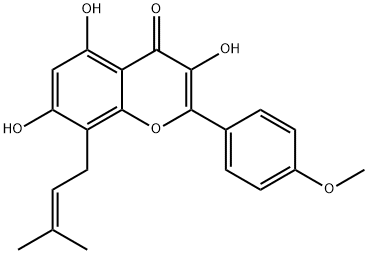
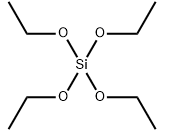
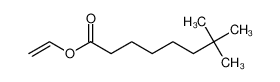
Pivaloyl chloride literature
Nickel-catalyzed direct C-H bond sulfenylation of acylhydrazines
Li, Jun-Ming,Yu, Yang,Weng, Jiang,Lu, Gui
, p. 6047 - 6056 (2018)
A Ni-catalyzed direct C-H bond sulfenylation of acylhydrazines was developed. The reaction used N-(pyridinyl)hydrazine as the bidentate-directing group, which can be smoothly removed through reductive N-N cleavage. This system can bear various important functional groups, providing an efficient route for the preparation of diverse diaryl sulfides.
-
Landis,Hamilton
, p. 1742 (1960)
-
REACTION OF SULFURYL CHLORIDE WITH P-TRIMETHYLSILYL-C-TERT-BUTYL-C-TRIMETHYLSILYLOXYMETHYLENEPHOSPHINE
Ionkin, A. S.,Nikolaeva, N. V.,Arbuzov, B. A.
, (1990)
-
-
Lennon,Stimson
, p. 7562 (1969)
-
AKT INHIBITOR
-
Paragraph 0122-0123; 0128; 096-0197, (2021/12/18)
The present invention discloses an AKT inhibitor, and specifically relates to a compound represented by formula I or a pharmaceutically acceptable salt thereof. The present invention further provides a preparation method thereof, and the use thereof in prevention and/or treatment of a disease mediated by AKT protein kinase.
Rhodium(III)-catalyzed chemodivergent annulations between phenyloxazoles and diazos via C–H activation
Zhang, Xueguo,Wang, Peigen,Zhu, Liangwei,Chen, Baohua
supporting information, p. 695 - 699 (2020/06/28)
Acid-controlled, chemodivergent and redox-neutral annulations for the synthesis of isocoumarins and isoquinolinones have been realized via Rh(III)-catalyzed C[sbnd]H activation. Diazo compounds act as a carbene precursor, and coupling occurs in one-pot process, where adipic acid and trimethylacetic acid promote chemodivergent cyclizations.
Synthesis, computational studies and enzyme inhibitory kinetics of benzothiazole-linked thioureas as mushroom tyrosinase inhibitors
Ujan, Rabail,Saeed, Aamer,Ashraf, Saba,Channar, Pervaiz Ali,Abbas, Qamar,Rind, Mahboob Ali,Hassan, Mubashir,Raza, Hussain,Seo, Sung-Yum,El-Seedi, Hesham R.
, p. 7035 - 7043 (2020/08/12)
Herein, we report synthesis of a set of benzothiazole-thiourea hybrids with aromatic and aliphatic side chains (BT1 to BT9) using an elegant synthetic strategy. The newly synthesized benzothiazole-thiourea conjugates were subjected to In-vitro tyrosinase inhibition and free radical scavenging activity. Majority of the compounds indicated inhibition considerably improved than the standard; compound (Kojic acid with IC50 = 16.8320 ± 1.1600 μM) BT2 with IC50 = 1.3431 ± 0.0254 μM was found to be the best inhibitor. A non-competitive mode of inhibition of BT2 was disclosed with Ki value of 2.8 μM. In order to study enzyme-inhibitor interactions SAR analysis molecular docking was carried out. The amino groups of thiourea were involved in hydrogen bonding with Glu322 showing the bond length of 1.74 and 2.70 ?, respectively. Moreover, the coupling of π-π was displayed between benzothiazole and benzene rings of His244 and His263, respectively. The outcome of this study might help to develop new inhibitors of melanogenesis, important for cosmetic and food products. Communicated by Ramaswamy H. Sarma.
Benzimidazole tethered thioureas as a new entry to elastase inhibition and free radical scavenging: Synthesis, molecular docking, and enzyme inhibitory kinetics
Abbas, Qamar,Ashraf, Saba,Channar, Pervaiz Ali,Hassan, Abbas,Hassan, Mubashar,Rafique, Hummera,Raza, Hussain,Rind, Mahboob Ali,Saeed, Aamer,Seo, Sung-Yum,Ujan, Rabail,Ul-Hamid, Anwar
, (2021/07/02)
The porcine pancreatic elastase inhibition and free-radical scavenging play a crucial role in age progression. All the series of 10 newly synthesized benzimidazole thioureas (4a-j) were assessed for elastase inhibition and radical scavenging activity to identify the suitable anti-aging ingredient for cosmetics products. The compounds 4e, 4f, 4g, and 4h showed inhibition better than the standard, while compound 4f showed the most significant elastase inhibition with the IC50 value of 1.318 ± 0.025 μM compared with oleanic acid IC50 13.451 ± 0.014 used ±1.989 and 41.563 ± 0.824, respectively, as standard. Molecular docking studies were performed and the compound 4f showed binding energy of 7.2 kcal/mol. Kinetics studies revealed inhibition of the pancreatic elastase in a competitive manner. The relative binding energy and structure activity relationship (SAR) identified compound 4f as an effective inhibitor of porcine pancreatic elastase. Compounds 4e and 4i showed remarkable free-radical scavenging activity with SC50 values of 26.421.